The ASPE Technical Expert Panel on Improving Cancer Policy Research through Information Technology
Submitted by
2987 Clairmont Road, Suite 220
Atlanta, Georgia 30329
100 Capitola Drive, Suite 104
Research Triangle Park, NC 27713
The views expressed in this paper are solely those of the authors, and do not necessarily reflect the views of the Department of Health and Human Services.
This report was produced under a contract to SciMetrika by the Office of the Assistant Secretary for Planning and Evaluation, Office of Science and Data Policy. The ASPE project Officer was James Sorace MD MS.
Contact Information
James Sorace MD MS
Senior Medical Officer
Assistant Secretary for Planning and Evaluation
Science and Data Policy
202-205-8678
| Today, cancer is one of the leading causes of death in the United States. The initial diagnosis of cancer is a complex process involving many healthcare specialists. Two physician specialties often at the center of this initial cancer diagnosis are radiology and pathology. While neither radiologists nor pathologists are usually involved in direct patient care, their collective findings and interventions are responsible for subsequent cancer patient treatment and outcome. The issue of workflows between radiology and pathology and the need for communication, correlation and resolution of discordant findings between the two groups to reduce diagnostic errors is the focus of this report. The report also discussed how tumor registries can benefit from these improved workflows. |
Executive Summary
Today, cancer is one of the leading causes of death in the United States. 1 The initial diagnosis of cancer is a complex process involving many healthcare specialists. Two physician specialties often at the center of this initial cancer diagnosis are radiology and pathology. While neither radiologists nor pathologists are usually involved in direct patient care, their collective findings and interventions are responsible for subsequent cancer patient treatment and outcome. A patient with radiology findings suggestive of malignancy is sent for biopsy to confirm or negate the diagnosis. These tissue specimens are sent to pathology where a definitive diagnosis is given based on the submitted material.
Unfortunately, not all institutions correlate suspicious radiology and subsequent negative pathology findings. As a result, there are instances when a radiologist issues a report stating a high degree of suspicion for malignancy, but, through sample or processing error, the material reviewed by a pathologist is not representative of the suspicious area detected in radiology. As a result, a negative for malignancy pathology report is issued. In order to avoid such false negative cancer diagnosis, it is imperative that both radiologists and pathologists communicate and correlate their findings in a way that ensures the proper treatment course for a patient. The workflow of reports originating in radiology and subsequently made available to pathology, as well as the reverse process, is essential to ensure that real time correlation of findings is maintained throughout the patient’s encounter. The issue of workflows between radiology and pathology and the need for communication, correlation and resolution of discordant findings between the two groups to reduce diagnostic errors is the focus of this report.
The Office of the Assistant Secretary for Planning and Evaluation (ASPE) commissioned SciMetrika to conduct a series of discussions with representatives from several groups to determine the current workflow practices between radiology and pathology and ways communications can be improved. The panel experts consisted of pathologists, radiologists, medical informaticists and cancer registrars involved in the delivery of healthcare or the study of the flow of health information.
In order to best illustrate the essential workflow needed to foster the relationship between radiologists and pathologists, the decision was made to study the processes involved in the diagnosis of breast cancer as a preeminent example. The panel focused on mammography studies, reviewing the roles and responsibilities of both radiologists and pathologists in the diagnosis of breast cancer and the current interactions between the two specialties. It became apparent through discussion and literature review that radiology and pathology often operate in different and distinct areas of a hospital or institution (or in differing institutions) with varying degrees of interaction. 2 This isolation increases the risk of radiologic-pathologist discordance, i.e., histologic findings that do not substantiate imaging findings. Reviewed studies assessed the frequency of radiologic/pathologic discordance of percutaneous breast biopsies from 1 percent to 6 percent. 3 These discordance rates have serious consequences if left unresolved. Based on a pilot study conducted by one panel member, we estimated that approximately 10,000 breast cancer cases may be under diagnosed per year due to the failure to resolve discordant radiology and pathology findings. It is obvious that any improvements in the process of communicating findings between radiology and pathology to ensure timely exchange of clinical information could reduce these false negative reports.
The panel developed a series of workflows to support optimal communication efforts to reduce discordant rates, not only for mammography studies, but also applicable across all cancer types. These workflows and the critical components of each stage are examined in detail. An idealized workflow model that not only includes radiology and pathology but also illustrates the importance of data flow between other disciplines is presented in this document. This report also addresses some of the limitations for the proposed solutions as well as the efforts of two institutions ( Kansas University Medical Center and the University of California Los Angeles) to resolve discordant findings between radiology and pathology. Included in these recommendations is the need to standardize the terminology of the reports to reduce misinterpretations as well as to expedite the flow of clinical information between radiology and pathology. This standardization is carried over to include the development of standardized requisition forms that accompany the specimen sent to pathology as well as the use of standardized messaging formats between information systems.
Both pathology and radiology organizations recognize the gaps in workflows that might hinder optimal communication of patient data, and progress is being made to resolve these critical issues and concerns. This report concludes with opportunities for both radiology and pathology to improve workflow processes that would improve communication and resolution of discordant findings. Suggestions include efforts to develop quality assurance programs and best practice guidelines that meet not only the needs of pathologists and radiologists but most importantly, provide optimal patient care.
1.0 Introduction
Malignant disease is one of the leading causes of death in the United States with over 11 million people with a cancer diagnosis. 1,4 Pathologists and radiologists are the physicians most often responsible for the initial diagnosis of cancer. While neither pathologists nor radiologists are generally considered providers of direct patient care, the information generated by these specialists is vital to patient diagnosis and subsequent selection of therapeutic options. Patients, surgeons and oncologists are the primary recipients of these reports, but there are other healthcare actors, such as clinical trial nurses, bio-repositories, insurance companies and local and national public health reporting agencies, which also use this information.
Despite being separate specialties, pathologists and radiologists are increasingly required to collaborate to diagnose and stage cancer cases. This relationship has grown in part due to modern imaging technology’s ability to detect and biopsy earlier, and therefore smaller, lesions as well as the need for both specialists to correlate their patient-specific findings. To increase and substantiate correlations, radiologists and pathologists require more open lines of communication including the creation of standardized, structured electronic reports that can be easily exchanged between the two specialties and with other physicians.
Because the patients’ health depends on these critical diagnoses, the Office of the Assistant Secretary of Planning and Evaluation (ASPE) sponsored a study to determine if the processes and workflows of radiologists and pathologists could be improved. To implement this study, SciMetrika, the contractor, convened a work group that included members with domain expertise in the fields of pathology, radiology and medical informatics. These individuals, including representatives from the Kansas University Medical Center (KUMC) and University of California Los Angeles (UCLA) contributed to the development of this report. The representatives from KUMC and UCLA were chosen as members of this group based on their active collaborative programs between the pathology and radiology departments at their respective institutions.
The work group was initially tasked with developing a consensus about the scope of collaborative efforts between these two specialties. In numerous meetings, the limitations of the current radiology-pathology workflow process and possible ways to improve it were discussed. The group also considered two alternative collaboration models adopted by KUMC and UCLA to integrate the work of radiologists and pathologists to improve communications. Both efforts stress the correlation of radiology and pathology results to improve the overall outcomes of the cancer diagnosis and treatment process. In the KUMC model, a multidisciplinary conference approach is used to resolve issues of radiologic-pathologic discordance, specifically in the diagnosis and treatment of breast cancer cases. At UCLA, a new project has been developed to establish integrated radiology-pathology workflows and reports for a number of malignant diseases. Both models will be discussed in later sections of this report.
2.0 Overview of Radiology and Pathology
In order to fully explain the issues discussed in this document, we must first explain the roles, responsibilities and interactions of radiologists and pathologists involved in patient care.
2.1 Radiology
There are currently more than 30,000 practicing radiologists in the United States. 5 Their practice involves a wide range of technologies that enable tumors to be detected, biopsied and tumor extent to be determined. Radiologists often specialize in either diagnostic radiology, which deals with the interpretation of images, or interventional/procedural radiology, which deals with providing therapeutic treatment for patients and often obtaining tissue biopsies. Breast imaging is a subspecialty of radiology that includes both diagnostic imaging such as mammography, ultrasound, and magnetic resonance imaging (MRI) as well as interventional procedures such as image-guided breast biopsies.
2.1.1 Roles and Responsibilities of Radiologists
Radiologists play a key role in cancer diagnosis and clinical staging. As imaging technology improves, radiologists are able to detect smaller and therefore earlier tumors. Increasingly, radiologists also obtain diagnostic tissue from the patient and send it to the laboratory along with pertinent clinical information for further analysis by a pathologist. Additionally, radiologists conduct a range of imaging studies that can detect the presence of cancer metastases. This information is used to determine the stage of a patient’s cancer. Finally, radiologists are responsible for releasing reports of their findings that become part of the patients’ medical record.
2.2 Pathology
There are about 17,000 pathologists in the United States. 6 Pathologists evaluate a specimen by direct macroscopic (visual examination with the unaided eye) and microscopic examination of tissues, and through the use of additional studies such as molecular pathology to render a definitive diagnosis of cancer. In addition, the pathologist determines tumor characteristics including organ of origin, histologic type, grade, pathologic stage, and other characteristics that determine prognosis and guide therapy.
2.2.1 Roles and Responsibilities of Pathologists
Pathologists play a pivotal and expanding role in the diagnostic process. They are responsible for drafting reports of their findings from tissue removed from patients by other physicians (e.g. radiologists, surgeons). Tissue samples vary from small biopsies such as image-guided needle biopsies to large surgical specimens. Once the specimen is received in the pathology laboratory, pathologists and their assistants grossly evaluate, process, and prepare slides for histologic microscopic evaluation. When needed, pathologists perform ancillary studies that help define diagnostic and therapeutic options. The pathology report, which becomes part of the patient’s medical record, is released to the patient’s referring clinician, who is responsible for informing the patient of the results.
To summarize, radiologists localize and characterize lesions through imaging studies, and may remove tissue for pathological diagnosis, while pathologists receive the tissue, sample it, and determine whether or not a malignant disease is present. Both radiologists and pathologists must rely on the adequacy of accompanying clinical information in rendering his or her diagnosis. Additionally, both specialists must create and release reports of their findings.
3.0 Description of the Problem
The successful diagnosis of malignancies depends on precise workflows that support the transfer of adequate and accurate information and tissue samples between the radiologist and pathologist. When the radiologist samples the correct location and the pathologist diagnoses malignant disease in the sample, the patient is typically referred to a cancer care team and the case is reviewed by a hospital-specific cancer committee. Today, radiologists can routinely detect smaller lesions based on radiologic findings such as microcalcifications. This process necessitates a close communication between the specialties to correlate findings. To give an example as to how small these findings can be, one study determined that microcalcifications with an average length of more than 0.41 mm were associated with a malignancy 77 percent of the time, while microcalcifications with an average size of less than 0.41 mm were associated with a malignancy in 29 percent of cases. 7 Smaller lesion size results in greater difficulty in sampling the lesion by both the radiologist and the pathologist. Often, the diagnostic evaluation by radiologists and pathologists occurs in separate departments with widely varying levels of collaboration. 2 This relative isolation increases the risk of radiologic-pathologic discordance. Radiologic-pathologic discordance occurs when the histologic findings do not correlate with or provide sufficient explanation for imaging results. 8
This issue of discordance can relate to either a false positive or false negative diagnosis. As noted above the case of patients with a pathology diagnosis of malignant disease is routinely reviewed by a multidisciplinary cancer committee, and there is a chance that a false positive error may be detected. However, discordance is especially problematic for suspicious radiologic findings with negative pathology results, as the information is not typically further evaluated by a committee or board and the possible health consequences for patients can be significant. These consequences include failure to recognize lesions that were inadequately sampled, false negative results with delayed diagnosis of malignancy, or failure to recognize non-malignant high risk lesions that should prompt additional testing or more aggressive clinical surveillance. Possible causes for discordant results include inadequate sampling of lesions (by either the pathologist or the radiologist), which signifies a need for further examination such as repeat biopsy or surgical excision of lesions 8, or the failure to recognize pre-malignant lesions that require further testing and/or careful monitoring.
4.0 Breast Cancer as an Object Lesson
4.1 Background
Due to complexities in radiology-pathology interaction in the diagnosis of cancer and the variations in the systems in place based on the type of cancer involved, the work group focused on one type of cancer as an object lesson. The group’s reasons for choosing breast cancer are outlined below.
4.2 High clinical volume and existence of extensive breast cancer guidelines
The number of mammograms performed annually in the US is estimated at approximately 36.7 million mammograms. 9 These mammograms result in approximately one million image-guided breast biopsies per year. 10-12 Current guidelines by the American College of Radiology (ACR) stipulate that radiologists are required to use standardized terminology such as Breast Imaging Reporting and Data System (BI-RADS®) under the Mammography Quality Standards Act (MSQA) of 1992 13 to help standardize mammography practice. This standard provides a clear set of terms and definitions for the breast imaging process and a mandatory set of final assessment categories to indicate level of certainty with respect to abnormal findings. It also includes a small set of actionable conclusions, oriented toward decision making with the highest level of certainty defined as Known Biopsy-Proven Malignancy-Appropriate Action Should Be Taken. Note that this highest level of certainty is dependent on the radiologist receiving confirmation of biopsy results. Breast cancer is unique in that the use of standardized terminology for unambiguous reporting of mammographic results such as BI-RADS is required by law under MQSA. MQSA guidelines also require that if a biopsy is performed, then radiologists must make an attempt to correlate the results of the biopsy with the mammogram findings. 13 This degree of specificity is not required in reporting the findings of other types of malignancies.
4.3 Availability of literature and data on breast cancer discordance
Several studies assessed the frequency of radiologic-pathologic discordance of percutaneous breast biopsies. In a study conducted at the Kettering Cancer Center in New York, Liberman et al. found that imaging-histologic discordance was present in 3.1 percent of examined lesions. 11 Similarly, Lee et al found a discordance rate of 7 percent between radiologist and pathologist findings. 14 Some studies evaluated discordance in benign biopsies. Mihalik et al. found that 2 percent of benign breast cases were discordant. 15 Overall, the rates of radiologic-pathologic discordance of percutaneous biopsy have usually ranged from 1 percent to 6 percent. 3
Moreover, discordance of radiologic-pathologic findings can have serious consequences in failing to properly diagnose carcinoma. 8,11 Further investigations of discordant cases play a significant role in identifying previously misdiagnosed or missed malignant lesions. In one study, one discordant case out of 25 benign cases was found to be malignant after open excision, giving a false-negative rate of 4 percent. 15 Liberman et al. identified carcinoma in 24.4 percent of discordant lesions after performing repeat biopsy. 11 Lee et al. detected cancer in 30 percent of discordant cases after subsequent surgical excisions, including cases of both ductal carcinoma in situ and invasive carcinoma. 14 Carcinoma was found in 11.1 percent of benign biopsies that underwent subsequent ultrasound-guided directional vacuum-assisted removal (DVAR) and 12.5 percent of cases that underwent surgical excisions. 3
In addition, unpublished data of a pilot study conducted at KUMC supports the published literature discussed above. Collaboration between radiologists and pathologists through multidisciplinary conferences at KUMC (additional information on KUMC’s multidisciplinary model discussed below) to resolve discordant benign cases helped identify missed cancer cases and changed the treatment plans for several patients. This data and how it was used to estimate the population-level impact of discordance are described below under the section discussing the public health impact of discordance.
Overall, the results of the published literature and unpublished data clearly indicate a significant problem with discordance and the importance of resolving any discordance between radiology and pathology findings. In some cases, this involves performing additional diagnostic studies at the time of pathologic examination.
Table 1 below shows six data items for which radiologic values are typically established before breast biopsy that need to be correlated with pathologic findings at the time of pathologic examination.
| Radiologic data item | Pathologic data item |
|---|---|
Calcification and pattern | Microcalcifications |
Tumor size | Tumor size |
Localizing wire | Wire location |
Contiguous involvement | Margins and contiguous involvement |
Number of discrete abnormalities | Number of discrete tumor foci |
Degree of clinical suspicion (e.g. BI-RADS score) | Final pathological diagnosis |
4.4 The KUMC Model: Techniques designed to reduce breast cancer discordance between pathology and radiology results
Multidisciplinary collaboration of radiology, pathology, and other involved disciplines to establish diagnostic concordance has been shown to impact clinical decision-making and decrease the rate of discordance overtime. 15 One such example of the utilization of multidisciplinary collaboration is the weekly cancer conferences adopted by KUMC.
In this innovative effort, radiologists and pathologists use audio-video conference technology to overcome hurdles such as manpower, time, and geographic constraints in order to conduct weekly multidisciplinary meetings. In these conferences, both groups view pathology findings and radiologic images simultaneously and consider findings and treatment recommendations. The specialists resolve any differences between radiology and pathology results and issue an integrated diagnostic report for each patient. The integrated report is completed and issued by the radiologist following the recommendations of ACR.
The KUMC project included a pilot study to determine whether this web interdisciplinary approach affected decision-making for treatment plans for patients with discordant radiologic-pathologic findings. The results of this study, presented at the Pathology Visions Conference (October 2008) and at the Molecular Summit 2009 (February 2009) revealed that web-based interdisciplinary conferences had a major impact on the clinical decisions made in one third of the benign discordant cases that were reviewed leading to re-excision of the lesion, re-biopsy, and/or additional radiologic follow-up.
4.5 The Availability of data that allows the public health impact of discordance to be estimated
Increased concordance of pathology results with imaging findings will not only change some individual diagnoses but will have an impact on population morbidity and mortality and may improve the quality and efficiency of cancer treatment. Although it is difficult to determine its exact impact, an estimation of the number of missed breast cancer diagnoses can be suggested.
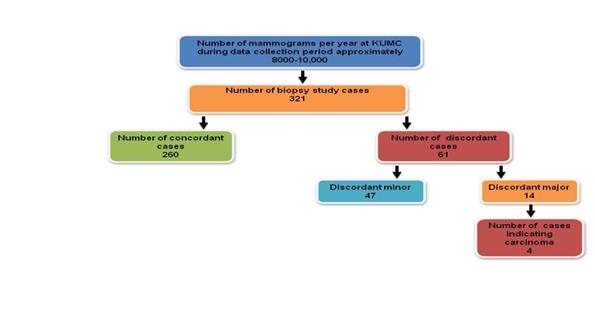
Based on the pilot study conducted at KUMC, 321 benign biopsy results were selected for radiology and pathology review. Of these 321 cases, 61 were found to have discordant results and upon further examination, four cases were found to be malignant. Using these results and projecting on a population level, (based on approximately one million needle biopsies performed per year in the US for the diagnosis of breast cancer with approximately 200,000 positive cases) 10-12,16 , we can estimate that approximately 9,969 missed cancer diagnoses per year ( Figure 1 ) could be treated earlier if radiology-pathology concordance processes were implemented nationwide. The American Cancer Society estimates that there will be 207,090 new breast cancer cases in 2010. 16 Hence, this group of missed or delayed diagnoses could represent about an additional 5 percent of these new cases.
We also completed these calculations using comparable data reported in a number of published studies. 3,17,18 The results of these calculations are reported in Table 2 . Although the results show a broad range of values, the group favors a value between 5,000 and 10,000 cases annually. However, additional studies are warranted in order to produce estimates with appropriate confidence levels.
Figure 1 : KUMC Pilot Study Results
| Author | Identified Malignant Cases | Benign Biopsy Cases | Estimated Missed Cases | Comments |
|---|---|---|---|---|
| Kim, 2007 | 3 | 634 | 3,785 | |
| Kim, 2008 | 2 | 386 | 4,145 | Note that an additional 11 malignancies were diagnosed in the biopsy negative non-discordant population. |
| Youk, 2010 | 28 | 1,706 | 13,130 | study found 50 malignant cases, but only 28 were due to imaging-histologic discordance |
4.6 The Applicability of most of the issues and proposed solutions associated with breast cancer to other cancer types
The work group determined that many of the problems identified in clinical scenarios involving the diagnosis of breast cancer were general issues applicable to the collaboration between the two specialties in the diagnosis of other cancers. These issues include developing workflows that support the:
- Communication of clinical information from the referring physician to both the radiologist and pathologist;
- Communication of clinical information from radiologist to pathologist including the current imaging study’s assessment of the risk of malignancy;
- Identification of specimens taken from patients by the radiologist;
- Receipt and processing of the sample by the anatomical pathology laboratory;
- Communication of results including negative findings from pathologists to radiologists;
- Achievement of radiology and pathology concordance and recommendations that discuss diagnosis and therapeutic options.
- Detailed descriptions of these important components, as well as limitations of current workflows follow.
5.0 Current Limitations and Proposed Solutions
5.1 Background
As discussed previously, a structured workflow that supports the transfer of relevant information and specimens between radiologists and pathologists can be crucial to the diagnosis of breast cancer cases as well as other types of cancer. This section discusses the critical components that must be supported by workflows, and addresses some of the limitations in the existing workflows of both radiologists and pathologists. This section also describes other efforts in radiology and pathology to address the needs associated with workflows.
5.2 Critical components of radiology-pathology workflows in diagnosis of cancer
Initially, a workflow must ensure the proper communication of pertinent clinical history including history of prior diseases. This patient information is usually sent from the referring clinician to the radiologist and/or pathologist. It is imperative that this clinical information be shared between radiologists and pathologists as both may not have received the information from the ordering clinician. It is also necessary that patient history be captured in the medical record as it might be the only source of patient information for either the radiologist or pathologist. In addition, a workflow must support the communication of the findings of any prior diagnostic imaging studies, including the degree of suspicion related to malignant disease and other relevant findings.
In order to ensure optimal patient care, information and specimen submission workflows should enable radiologists and pathologists to unambiguously link a pathology specimen to the following information:
- The demographic and clinical history of the patient;
- Contact information to facilitate communication between the radiologist and pathologist;
- Information related to the appropriate recipient of the final report;
- Pertinent data on the specimen itself, including
a. The anatomical origin of the specimen;
b. The methods used in the collection and pre-analytical processing prior to arrival at the pathology laboratory;
c. Unique specimen identifiers (e.g., bar codes) that can be used to securely label the samples at the time and site of collection.
Workflows must ensure the flow of communication of results (both positive and negative) between pathologists and radiologists to support the formal correlation process of radiology and pathology results and resolve any discordance in a timely manner. The proposed workflow in Figure 2 illustrates a high level summary of an idealized workflow model that maximizes the information transfer between the primary disciplines under consideration and identifies key roles including the referring clinician, radiologist, and pathologist in addition to other users of radiology-pathology information like cancer registries. Appendix A presents more detailed illustrations of each phase of the workflow process – screening, biopsy, and diagnosis.
Figure 2 : Proposed Workflow
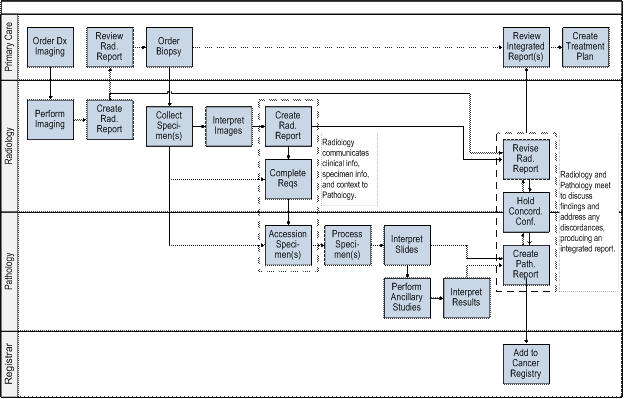
This diagram represents a high-level summary of an idealized workflow process that maximizes the flow of information between the referring clinician, radiologist, and pathologist involved in the cancer diagnosis process as well as additional users of radiology-pathology information such as cancer registries.
Ultimately, the patient’s medical records must include not only clinical history but also a complete account of all the relevant radiology and pathology information, preferably in an electronic format.
5.3 Limitations to current workflows
Current attempts to address the radiology-pathology workflow needs are being carried out in isolation by some institutions but there is no unified approach to addressing these recommendations. There are multiple issues that need to be addressed and resolved before a unified workflow can be adopted. First, there are numerous different, unique information systems used by the two disciplines. Even within pathology, anatomical pathology and laboratory medicine (clinical pathology) might have separate information systems that need to interoperate with each other as well as with other systems such as Hospital Information System (HIS) and Electronic Medical Records (EMR). Also, a separate pathology system may be needed to view and possibly store pathology images. Similarly, radiologists typically use a Picture Archiving and Communication System (PACS) to communicate and archive images, a separate Radiology Information System (RIS) for textual reports, and often a third system for speech recognition. In addition, a separate system is often used in radiology for only mammography studies [*] (Mammography Information System (MIS)). For most intra-institutional communications between radiology and pathology to occur, the radiology and pathology systems need to communicate with the main HIS.
Second, there is lack of standardization related to the exchange of information between the various pathology and radiology information systems and where an exchange has been developed implementation is typically incomplete. Both pathology and radiology information systems may use a messaging standard such as Health Level Seven (HL7) for orders and results. However, current implementations do not support the advanced HL7 features needed to support ordering or reporting functions for standardized interfacing between radiology and pathology information systems. There is also a lack of commercially available information systems that allow a unified view of a patient’s records without querying across multiple systems to obtain relevant information from a radiology or pathology system. This situation is even more complicated in settings where the pathologist and radiologist do not share a common host institution and/or HIS.
Third, while there may be general mechanisms for standard communication of specimen-associated data from the biopsy environment to the Laboratory Information System (LIS), there is no national standardized electronic specimen requisition form. Instead, a paper requisition form accompanies the physical specimen sent to the pathology laboratory from radiology and is used to communicate clinical and specimen information.
Finally, while there are standardized protocols (such as the College of American Pathologists Cancer Protocols) to report malignant findings, there is no national consensus or guideline for the reporting of benign or negative pathology results. This can be problematic, especially if benign pathology results are discordant with radiology findings and may potentially be missed malignancies. Practice guidelines are needed for these types of cases to assist in the identification of possible discordant pathology/radiology results. By using standardized data elements, biopsy-imaging correlation may improve and the process may be automated if the data elements can be captured in a structured, machine-readable manner.
5.4 Progress towards addressing the needs of radiology and pathology workflows
Both specialties recognize that there are gaps in their respective workflows in terms of optimal communication and discordance resolution. Each specialty is taking steps to address these issues and limitations. In pathology, there are standardized cancer reporting templates developed by the CAP Cancer Committee that may require additional modifications to report negative biopsy results. In addition, there are current efforts within the pathology field to adopt standardized pathology terminology. This effort has been jointly developed by the CAP Cancer committee and the Pathology Electronic Reporting Taskforce (PERT), and was funded in part by the Centers for Disease Control and Prevention National Program of Cancer Registries (CDC NPCR). Several pathology vendors have begun incorporating these eCC templates into their reporting systems. 19
The radiology community has also undertaken similar endeavors in terms of standardized reporting and transmission of results. Beginning in 1983, ACR and National Electrical Manufacturers Association (NEMA) published a set of standards for the format and transmission of digital images that have come to be known Digital Imaging and Communications in Medicine (DICOM). The implementation of DICOM standards for medical images is nearly universal in radiology systems. Additional efforts that seek to address the limitations of reporting in radiology are underway. The Radiological Society of North America (RSNA) has released a comprehensive lexicon of radiology terms including procedure names called RadLex. 20 This tool extends the use of consistent terminology across radiology subspecialties. 20,21 RSNA is also developing structured reporting templates for common procedures in all radiological subspecialties. These report templates are designed to integrate all evidence collected during the imaging procedure including clinical data, coded terminology, technical parameters, measurements, annotations, and key images. 22 The templates need to be further modified and tested for the purpose of documenting biopsies and submitting specimens to pathology.
In addition to the progress made within each field, individual academic medical centers such as KUMC (discussed previously) are beginning to develop programs that facilitate the collaboration between the two specialties in order to improve patient care outcomes. Another example of the collaboration between the two specialties is the UCLA model that is discussed in detail in the next section.
5.5 UCLA’s integrated radiology-pathology project
5.5.1 Background
The UCLA model expands the KUMC model to incorporate a broader range of cancer types and diagnostic modalities. The UCLA Radiology-Pathology project, which is currently under development, is a joint venture by the UCLA departments of Radiology and Pathology to develop a single integrated report that encompasses both diagnostic modalities for evaluation of a mass lesion. This format will combine diagnostic imaging reports with image-guided biopsy reports and detailed pathology reports that will contain pertinent clinical staging information for patients with newly diagnosed tumors in solid organs.
To accommodate the goals of the report, including inclusion of additional diagnostic modalities such as molecular testing for prognosis and response prediction, the report will be electronic and web-based. This electronic report will allow the end-user to access the different and specific aspects of the diagnostic studies in one place without the need to wade through nonessential information. The report will also have a print report tab that will allow the entire report to be printed as a paper document for inclusion in the hard copy medical record.
5.5.2 Workflow of the integrated UCLA model
The report will be initialized by the interventional radiologist performing the biopsy. The radiologist will be responsible for linking the most appropriate diagnostic radiological study and radiological procedure with the pathology specimen. This will ensure that the end-user will have access to the most relevant radiological studies when reviewing the pathology findings. The radiologist will flag and annotate pertinent images from the diagnostic radiology study for the Rad-Path report, and he or she will sign-out the radiology study which will also exist as a distinct radiology report in the hospital medical record.
To assist the radiologist and ensure adequate sampling, a cytotechnologist will be available in the radiology suite to perform adequacy checks on the specimen, partition the specimen as needed into appropriate specimen containers, and accession the specimen into the pathology laboratory information system. The pathology specimen accession number and the Rad-Path report number will be included within the body of the radiology procedure report.
The specimen will be processed in the pathology laboratory according to normal routine. The pathologist will sign-out the specimen as per usual and the normal pathology report will be included in the hospital medical record. The pathology report will also include the Rad-Path accession number within the body of the report.
The Rad-Path report will be a separate report within the patient’s medical record that integrates information from the radiology and pathology reports and exists as an electronic document. To create the report, a program has been developed that processes HL7 data that is streamed from the pathology LIS system and radiology RIS system. To allow for automatic extraction, the radiology and pathology reports are specifically structured to facilitate retrieval of specific data from the reports to be inserted in the appropriate place in the Rad-Path report.
The pathologist and radiologist will communicate directly in person or by telephone to ensure appropriate coordination between the different diagnostic modalities for a unified report with correlated diagnostic results. This will alert the pathologist to potential discordance between the radiology findings and the pathology report before the final pathology report is issued. In doing so, the pathologist will be able to address the discordance in the pathology report, prevent diagnostic errors, or recommend further diagnostic testing before a final diagnosis is given to the patient.
5.5.3 Report format
Utilizing an electronic web-based report will enable many sections of the report to be collapsible into tabs to shrink the size of the report and reduce clutter (e.g. gross description, microscopic description, biopsy technique). Some ancillary studies, such as molecular diagnostic tests, may take several days to finalize. At that time, the results will be put into the front of the report and accessible by clickable tabs. All studies that are in progress will be highlighted pending. This process will enable the treating clinician to know exactly what tests have been ordered and the status of the tests. This will reduce unnecessary searches to see if tests have been ordered and save time. To notify the treating clinicians that Rad-Path report, shown in Appendix B, is finalized an email will be automatically sent to the ordering clinician with a link to the secured website for user login to view the results.
5.6 Limitations of the KUMC and UCLA models
In both the UCLA and KUMC case studies, the collaborating departments of radiology and pathology belong to a common parent institution (i.e. UCLA or the University of Kansas). However, in places where pathology and radiology are not housed in the same institution a more complex model exists and would require greater efforts to reduce the barriers that impede collaboration between radiologists and pathologists. However, both the KUMC and UCLA efforts present a useful starting point to improving the information systems, workflows, and ultimately patient-care models at the national level.
6.0 Benefits to Public Health and Research
6.1 Background
Although information collected in radiology and pathology systems is utilized by a number of different users for several purposes, this section will focus only on cancer registries because of their important role in the collection and dissemination of cancer data in the US . Cancer registries collect data produced by radiologists and pathologists on all cancer types that can be used to inform stakeholders such as public health authorities and researchers. Hence, the benefits of better integrated and collaborative radiology-pathology workflows and diagnostic reports have the potential to better support these activities. This section provides an overview of cancer registries and describes how they can particularly benefit from improved radiology-pathology workflows.
6.2 Cancer registries
Cancer registries provide information to clinicians and other healthcare providers, public health officials, administrators, and scientists for a variety of purposes. Their ultimate goal is to prevent and control cancer, including improvement of cancer patient care. 23 Since 1956, the American College of Surgeons has required all approved cancer programs to establish a cancer registry which can “measure the quantity and quality of medical care provided for cancer patients at a given institution.” Further, “the hospital cancer registry has been described as the “mirror” which can reflect to the hospital staff how well it is diagnosing and treating cancer in its hospital”. 24 The American Cancer Society expanded on this by enumerating four basic activities: 25
- To provide quality control information on the diagnosis and treatment of cancer.
- To perform follow-up processes to accumulate time-mortality data for assessing end results.
- To provide education of clinicians in the diagnosis of cancer.
- To provide a repository of bio-statistical data for research.
Today’s hospital cancer registry retains this description 45 years later, adding to its activities support for administrators, cancer control, and other public health programs.
Central cancer registries collect cancer information for the population residing in a defined-geographic location, usually a state or province. The uses of central cancer registry data include: 26
- Assessment of disease occurrence using rates;
- Calculation and assessment of survival estimates;
- Epidemiologic research and program support; and
- Cancer control activities
In addition to state-level cancer registries, federal/national cancer registry programs also exist to monitor the burden of cancer in the county/nation.
In the mid-1990s the cancer registry community, consisting of central and hospital registry standard setters, developed data standards for the electronic transmission of cancer abstract reports. These data standards allow for the electronic transmission of cancer abstract reports between the different registry software actors, e.g. hospital and central cancer registries, and national cancer registry programs. The data standards are updated on an annual basis as new data items and value sets are introduced and existing ones are modified or deprecated.
Cancer registries rely heavily on the anatomic pathology laboratory as it serves as the central source for identifying and confirming reportable cancer cases. Additionally, the pathology report is the primary medical report that provides the majority of the diagnostic information about the patient’s cancer. The most important information in the pathology report includes the source of the specimen, primary site, size of the tumor, histologic type and the grade of cancer, and stage/extent of disease progression. The results of additional clinical testing such as cytogenetics and molecular markers may also be provided within the pathology report. Cancer registries have developed standards for electronically transmitting pathology reports to a cancer registry, making the information available to the registry and its users in near real-time. 27,28 The cancer registry community has also developed standards for the interpretation of pathology reports where the degree of suspicion or level of certainty is not clearly apparent. Diagnostic terminology or modifiers are defined as being either considered as diagnostic of cancer or not. For example, a cancer which is described as presumed or probably cancer is considered diagnostic of cancer (reportable), while a diagnosis with the terms possible or suggestive of is not. 29 While the cancer registry community has a consistent standard for interpreting the degree of suspicion, radiologists and pathologists did not contribute to this standard and do not use it in their practice.
The diagnostic radiology/imaging department also serves as an important source in providing information to the cancer registry as radiologic procedures (e.g. x-ray, CT and bone scans) complement the pathology report in order to provide a more complete picture of the extent of cancer progression (stage) in the patient.
6.3 Challenges to cancer surveillance
One of the challenges to the existing cancer surveillance system is the amount of time and resources needed to identify reportable cancers/tumors and to collect the required information. One part of this challenge is the cancer surveillance system reliance on manual retrieval of information from the medical records (paper-based or electronic), containing the pathology and radiology reports, and the manual entry (or re-entry) of the salient information into the cancer registry database (another software system). The readiness and ability of cancer registry systems to capture information on new diagnostic (e.g. molecular markers) and treatment tools and protocols is also impacted by the over-reliance on this manual process.
An additional challenge that has not been mentioned as frequently is the amount of time cancer registrars spend reviewing and adjudicating information found within reports. A difference in documentation between radiology and pathology reports requires close attention to the rules for collecting the related data item and frequently requires a follow-up to both departments to clarify the discrepancy or ambiguity.
6.4 Opportunities to improving the work of cancer registries
Several opportunities to improve cancer registries’ ability to collect pathology laboratory information in a timely manner are underway. Synoptic pathology reports in an electronic format provide a means to automatically populate the cancer registry database, reducing the time spent in re-typing information, eliminating transcription errors, and more accurately capturing the intent of the pathologist. 30 However, what has not been address by the cancer registry community is the challenge of discrepant or cross-report ambiguity between the radiologic and pathologic reports. An integrated workflow process that ensures the flow and availability of complete information produced by the two departments would improve the quality of cancer registry data including the accurate identification of potentially missed cancers, enhance the effectiveness of cancer registries operations, and conserve limited resources.
7.0 Additional Opportunities and Conclusions
Improvements to the collaborative workflow processes between radiologists and pathologists are critical in cancer diagnoses. Implementing a system or process that allows optimal communication exchange between radiology and pathology will provide immediate benefits for practitioners and patients. It will also provide other users of radiology and pathology information, including cancer surveillance initiatives with more accurate and complete data on the status of cancer at the state and national level. Unfortunately, there is often little to incentivize institutions to develop the necessary infrastructure for radiology and pathology systems to share information with each other. Considering that the two specialties have different member organizations, there is currently no organizational home to host the interoperability or drive the different systems to interoperate.
There are several opportunities to consider for improving the current radiology-pathology workflow processes and systems. As previously discussed, radiology and pathology information systems must support the workflows required for the specialties to exchange specimen findings and ensure clinical concordance on each case. In a recent survey by CAP, 88 percent of responding pathologists indicated that they would like to review radiologic images. 31 Achieving this will require continued refinement of electronic standards such as CAP’s Cancer Protocols and Checklists and RSNA’s Report Templates to support the interoperability between pathology and radiology systems that allow access to each other’s findings. The group also noted that while the DICOM standards for image exchange in radiology have been in the market for several years, the DICOM supplement for digital pathology (Supplement 145) [†], has only recently been released (July 2010). 32 While it will take commercial developers some time to implement this standard into their systems, it creates an opportunity for the market to develop products specifically designed to meet the needs of this new collaborative model. To further support these needs, the workgroup identified four gaps in current standards efforts that should be addressed in the future.
- The need to develop ordering/requisition standards that enable radiologists to uniquely identify tissue samples and link them to the associated clinical information for submission to the pathology laboratory
- The need to develop appropriate linkages between documents so that the appropriate information can be reviewed during a concordance process
- Paradoxically, the need to develop standardized reporting templates that pathologists can use to report non-malignant findings
- The need to establish a mechanism to document concordance between radiology and pathology results
A second potential opportunity to improve radiology-pathology workflow systems is to view the interaction as a model for an episode of patient care with reimbursement components that could foster concordance meetings and collaborative quality improvement programs between the specialties. It is important that these models enable a wide range of partners to interact both within an institution and between institutions. The group noted that electronically reporting the results of diagnostic studies to cancer registries could be the first step in developing these models as it would foster the use of standardized reporting templates that are already available while offering the tumor registries the benefits of more timely reporting of cancer incidence.
Another opportunity is for the two specialties to foster educational exchanges between the disciplines and expose both practitioner types to training in the other discipline. These efforts should be implemented at the medical school, post-graduate medical education, and continuing medical education levels. Recently, ACR launched the American Institute for Radiologic Pathology (AIRP). The institute will offer a Radiologic-Pathologic Correlation course beginning in January 2011. 33 This course will provide radiology residents with an opportunity to learn the corresponding pathologic counterpart of their findings and emphasize the correlation between pathology and the medical images. 33
In this analysis we have focused primarily on breast cancer as a model. However, the two specialties are also collaborating in many other types of malignancies. Future studies should examine the role of the radiology-pathology workflow processes in the broader clinical setting. These studies should include efforts to quantify both the rate of discordant findings and their public health effects. Their results should be used to develop dedicated quality assurance programs including quality measures and best practice guidelines that address the needs of pathologist, radiologist, and the patients they serve.
References
1. Xu J, Kochanek KD, Murphy SL, Tejada-Vera B. Deaths: Final Data for 2007. National Vital Statistics Reports. Vol 58. Hyattsville, MD: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics; 2010.
2. Raab SS, Grzybicki DM. Quality in Cancer Diagnosis. CA Cancer J Clin. 2010;60(3):139-165.
3. Kim M, Kim E-K, Lee J, et al. Breast lesions with imaging-histologic discordance during US-guided 14G automated core biopsy: can the directional vacuum-assisted removal replace the surgical excision? Initial findings. Eur Radiol. 2007;17(9):2376-2383.
4. Altekruse SF, Kosary CL, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2007. Bethesda, MD: National Cancer Institute; 2010.
5. American College of Radiology. About US. 2010; http://www.acr.org/MainMenuCategories/about_us.aspx. Accessed August, 20, 2010.
6. College of American Pathologists. CAP Facts. 2010; http://www.cap.org/apps/cap.portal?_nfpb=true&cntvwrPtlt_actionOverride=%2Fportlets%2FcontentViewer%2Fshow&_windowLabel=cntvwrPtlt&cntvwrPtlt%7BactionForm.contentReference%7D=media_resources%2Fcollege_facts.html&_state=maximized&_pageLabel=cntvwr. Accessed August 20, 2010.
7. Buchbinder SS, Leichter IS, Lederman RB, et al. Can the size of microcalcifications predict malignancy of clusters at mammography? Acad Radiol. 2002;9(1):18-25.
8. Youk JH, Kim E-K, Kim MJ, Lee JY, Oh KK. Missed Breast Cancers at US-guided Core Needle Biopsy: How to Reduce Them. Radiographics. January 2007 2007;27(1):79-94.
9. IMV. 2008 Mammography Benchmark Report. Des Plaines, IL: IMV;2008.
10. Liberman L, Gougoutas CA, Zakowski MF, et al. Calcifications Highly Suggestive of Malignancy: Comparison of Breast Biopsy Methods. Am. J. Roentgenol. July 1, 2001 2001;177(1):165-172.
11. Liberman L, Drotman M, Morris EA, et al. Imaging-histologic discordance at percutaneous breast biopsy. Cancer. 2000;89(12):2538-2546.
12. Liberman L. US-guided core breast biopsy: use and cost effectiveness. Radiology. 1998;208(3):717-723.
13. U.S. Food and Drug Administration. Mammography Quality Standards Act (MQSA). 2010; http://www.fda.gov/Radiation-EmittingProducts/MammographyQualityStandardsActandProgram/Regulations/ucm110823.htm. Accessed August 19, 2010.
14. Lee J-M, Kaplan JB, Murray MP, et al. Imaging Histologic Discordance at MRI-Guided 9-Gauge Vacuum-Assisted Breast Biopsy. Am. J. Roentgenol. October 1, 2007 2007;189(4):852-859.
15. Mihalik JE, Krupka L, Davenport R, Tucker L, Toevs C, Smith RS. The rate of imaging-histologic discordance of benign breast disease: a multidisciplinary approach to the management of discordance at a large university-based hospital. Am J Surg. 2010;199(3):319-323.
16. American Cancer Society. Cancer Facts & Figures 2010. Atlanta, GA: American Cancer Society, Inc.;2010.
17. Youk J, Kim E-K, Kim M, Kwak J, Son E. Analysis of false-negative results after US-guided 14-gauge core needle breast biopsy. Eur Radiol. 2010;20(4):782-789.
18. Kim M, Kim E, Park S, et al. Imaging-histologic discordance at sonographically guided percutaneous biopsy of breast lesions. Eur J Radiol. 2008;65(1):163-169.
19. The Altarum Institute. Electronic Reporting in Pathology: Requirements and Limitations. Washington, DC: Office of Assistant Secretary of Planning and Evaluation, U.S. Department of Health and Human Services 2009.
20. Radiological Society of North America. RSNA Informatics: RadLex. 2010; http://www.rsna.org/informatics/radlex.cfm. Accessed May 20, 2010.
21. Langlotz CP. RadLex: A New Method for Indexing Online Educational Materials. Radiographics. 2006;26(6):1595-1597.
22. Radiological Society of North America. RSNA Informatics: Reporting. 2010; http://www.rsna.org/informatics/radreports.cfm. Accessed May 20, 2010.
23. Menck H. Types and Charactersitics of Central Registries, Ch 1 In: Hutchison C, Menck H, Burch M, Gottschalk R, eds. Cancer Registry Management: Principles and Practice. Second ed. Dubuque, IA: Kendall Hunt Publishing Co.; 2004.
24. Schneider A. Progress in Cancer Control through Cancer Registries. CA Cancer J Clin. 1958;8:207-210.
25. American Cancer Society. The Hospital Cancer Registry: Definition, Purpose, Value, Operation and Cost. New York, NY: American Cancer Society, Inc.;1966.
26. IntleKofer R, Michaud F. The National Program of Cancer Registries, Chapter 29 In: Menck H, Deapen D, Phillips J, Tucker T, eds. Central Cancer Registries: Design, Management and Use. Second ed. Dubuque, IA: Kendall Hunt Publishing Co.; 2007.
27. North American Association of Central Cancer Registries. Standards for Cancer Registries, Volume V, Pathology Laboratory Electronic Reporting, Version 2.0. Springfield, IL: North American Association of Central Cancer Registries Inc.;2006.
28. North American Association of Central Cancer Registries. Electronic Pathology (E-Path) Reporting Guidelines. 2006; http://old.naaccr.org/filesystem/pdf/E-Path%20Reporting%20Guidelines_FINAL_01-29-07%20.pdf. Accessed May 31, 2010.
29. North American Association of Central Cancer Registries. Standards for Cancer Registries, Volume II, Data Standards and Data Dictionary, Fifteenth Edition, Version 12.1. Springfield, IL: North American Association of Central Cancer Registries;2010.
30. U.S. Department of Health and Human Services. Report on the Reporting Pathology Protocols for Colon and Rectum Cancers Project. Atlanta, GA: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion;2005.
31. American College of Pathologists. Diagnostic Intelligence & Health Information Technology (DIHT) June 2009 Survey;2009.
32. National Electrical Manufacturers Association. Digital Imaging and Communications in Medicine (DICOM) Supplement 145: Whole Slide Microscopic Image IOD and SOP Classes. 2010; ftp://medical.nema.org/medical/dicom/2009/. Accessed October 4, 2010.
33. American College of Radiology. ACR Launches American Institute for Radiologic Pathology. 2010; http://www.acr.org/HomePageCategories/News/ACRNewsCenter/ACRLaunchesAIRP.aspx. Accessed July, 21, 2010.
Appendix A: Phases of the Proposed Workflow
Phase I: Screening
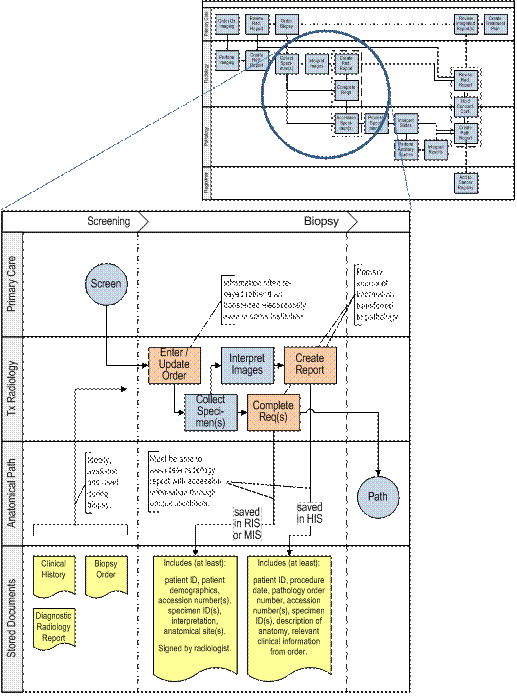
This diagram describes the interaction and flow of patient information between the referring clinician ordering the diagnostic study and the diagnostic radiologist.
Phase II: Biopsy

This figure demonstrates the biopsy process and the key responsibilities of the interventional radiologist including receipt of order for biopsy, collection of the biopsy specimen, interpretation of images, creation of a report of findings, and completing a requisition form to submit with the specimen for processing by pathologist.
Phase III: Diagnosis
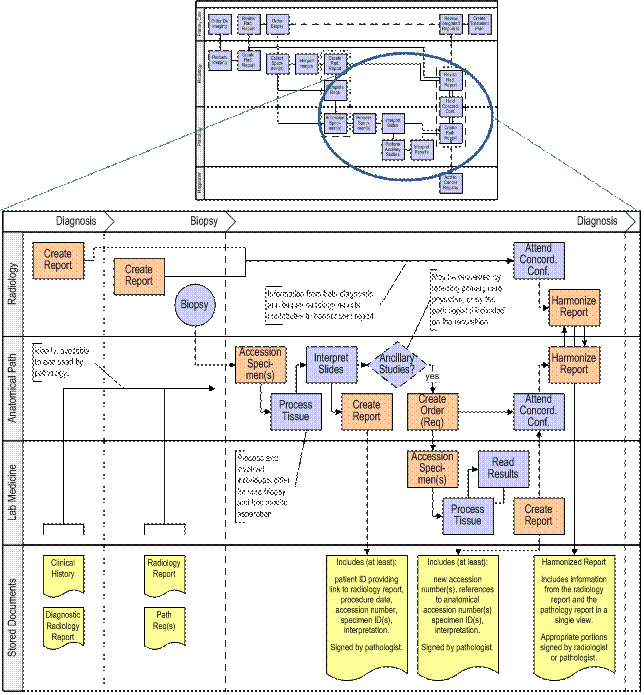
This diagram illustrates the interaction and workflow process between the radiologist and pathologist during the diagnosis phase starting with the requisition and accessioning steps through the completion of a harmonized concordant report that can be shared with the cancer registry.
Appendix B: UCLA Integrated Radiology-Pathology Summary

This figure is a template of how the electronic web-based UCLA integrated radiology-pathology report will look. It illustrates the main components of the report including patient information, integrated radiology-pathology summary, images, and additional studies.
[*] The Mammography Information System (MIS) is unique to the needs of mammography and stores data as B-RADS® categories
[†]This supplement will become part of the DICOM PS 3 Standard, which is under continuous maintenance by National Electrical Manufacturers Associations (NEMA). The current official version of the standard is available at ftp://medical.nema.org/medical/dicom/2009/
